
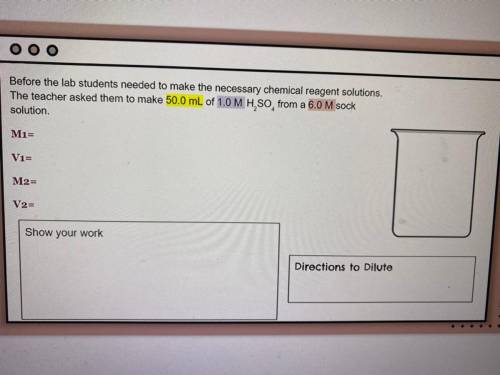
Before the lab students needed to make the necessary chemical reagent solutions, the teacher asked them to make 50.0 mL of 1.0 M H2SO2 from a 6.0 M sock solution
M1 =
V1 =
M2 =
V2 =
(Please help I had surgery so I didn’t even learn this lesson and literally none of my friends know either)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2

Chemistry, 23.06.2019 01:00
Which of the following is the molecular formula for a simple sugar? a. cooh b. h2o c. oh d. c6h12o6
Answers: 1
You know the right answer?
Before the lab students needed to make the necessary chemical reagent solutions, the teacher asked t...
Questions



Social Studies, 22.01.2021 23:40

Chemistry, 22.01.2021 23:40

Mathematics, 22.01.2021 23:40



Mathematics, 22.01.2021 23:40





Mathematics, 22.01.2021 23:40

History, 22.01.2021 23:40



Mathematics, 22.01.2021 23:40


Mathematics, 22.01.2021 23:40



