

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 20:30
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
You know the right answer?
An infant acetaminophen suspension contains 80 mg/0.80 mL suspension. The recommended dose is 15 mg/...
Questions



English, 01.04.2020 00:24


English, 01.04.2020 00:24

English, 01.04.2020 00:24


History, 01.04.2020 00:24

Social Studies, 01.04.2020 00:24




Health, 01.04.2020 00:24

History, 01.04.2020 00:24

Mathematics, 01.04.2020 00:24

Mathematics, 01.04.2020 00:24


Mathematics, 01.04.2020 00:24


Biology, 01.04.2020 00:24

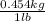 = 5.902 kg
= 5.902 kg

