8. A student collects the following data in their lab.
Trial
Temperature
Reaction Rate<...

Chemistry, 10.06.2021 20:30 giusto1073
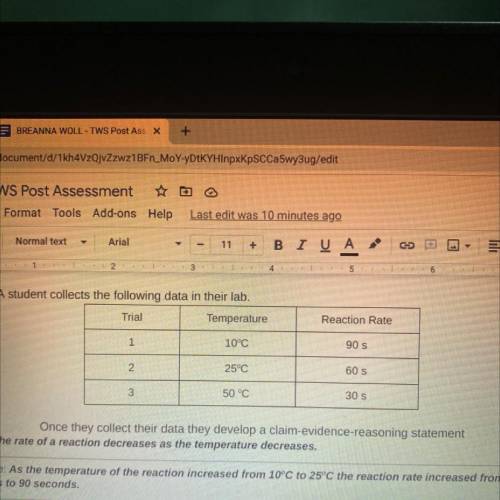
8. A student collects the following data in their lab.
Trial
Temperature
Reaction Rate
Once they collect their data they develop a claim-evidence-reasoning statement
Claim: The rate of a reaction decreases as the temperature decreases.
Evidence: As the temperature of the reaction increased from 10°C to 25°C the reaction rate increased from 60
seconds to 90 seconds.
Reasoning: Reaction rate depends on the temperature of the reactants. So when the temperature decreases, so
should the reaction rate. Particles need to collide in order to react. Lower temperatures allow molecules to move
slow enough to stick together and react so the reaction rate decreases.
Use the space below to provide feedback to the student. Was their conclusion correct? How should they fix it?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3
You know the right answer?
Questions



Spanish, 22.04.2020 20:29




Biology, 22.04.2020 20:29


Chemistry, 22.04.2020 20:29

Mathematics, 22.04.2020 20:29


English, 22.04.2020 20:29

History, 22.04.2020 20:29


Mathematics, 22.04.2020 20:29



Mathematics, 22.04.2020 20:29

Mathematics, 22.04.2020 20:30



