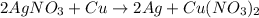Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 23:00
What is the oxidation state of each individual carbon atom in c2o42−?
Answers: 1

Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1

Chemistry, 23.06.2019 01:30
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2
You know the right answer?
__AgNO3 + __Cu → __Ag + __Cu(NO3)2...
Questions




Engineering, 16.01.2021 21:10

Biology, 16.01.2021 21:10


Mathematics, 16.01.2021 21:10

Mathematics, 16.01.2021 21:10






Mathematics, 16.01.2021 21:10




Mathematics, 16.01.2021 21:10

Physics, 16.01.2021 21:10

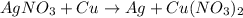
 = 1Cu = 1
= 1Cu = 1 by 2 on reactant side and multiply Ag by 2 on product side. Hence, the equation can be re-written as follows.
by 2 on reactant side and multiply Ag by 2 on product side. Hence, the equation can be re-written as follows.