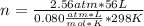Chemistry, 09.06.2021 08:50 bbenaventbbbb9653
Enough nitrogen must be generated in the bag to create a total pressure of 2.56 atm, which then drops as the bag slowly deflates. Assuming the volume of the bag is 56.0 L and the temperature in the car is 25oC, calculate the molar quantity (number of moles) of N2 that must be generated. Hint: Use Ideal gas law PV=nRT (Ideal gas constant R=0.080 L. atm/mol. K)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3


Chemistry, 22.06.2019 07:00
6what is the importance of water on earth? a) it keeps the top layer of the geosphere cool b) it allows life to exist c) it provides ice at the poles d) it creates earth's blue color from space
Answers: 2

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2
You know the right answer?
Enough nitrogen must be generated in the bag to create a total pressure of 2.56 atm, which then drop...
Questions

History, 02.12.2019 05:31

English, 02.12.2019 05:31

Mathematics, 02.12.2019 05:31



English, 02.12.2019 05:31




English, 02.12.2019 05:31

Mathematics, 02.12.2019 05:31


Mathematics, 02.12.2019 05:31

English, 02.12.2019 05:31



Mathematics, 02.12.2019 05:31

Mathematics, 02.12.2019 05:31

Mathematics, 02.12.2019 05:31

History, 02.12.2019 05:31

 T= 25 C= 298 K (being 0 C= 273 K)
T= 25 C= 298 K (being 0 C= 273 K)