
Chemistry, 08.06.2021 16:40 AbigailHaylei
4Fe (s) + 3 O2 (g) → 2 Fe2O3 (s)
A. What is the oxidation number for each item in this equation?
B. What is being oxidized?
C. What is being reduced?
D. What is the oxidizing agent?
E. What is the reducing agent?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:20
Asolution contains 180 g of glucose (c6h12o6) and 162 g of water. what is the mole fraction of glucose?
Answers: 3

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 22.06.2019 23:30
If it is an isoelectronic series select true, if not select false. o2-, s2-, se2-, te2- na+, k+, rb+, cs+ n3-, p3-, as3-, sb3- ag, cd+, sn3+, sb4+ f-, cl-, br-, i- f-, ne, na+, mg2+ s2-, s, s6+
Answers: 1

Chemistry, 23.06.2019 15:30
How is the electron sea model of metallic bonding different from the band theory? how are they the same? give at least one similarity and one difference between the models
Answers: 3
You know the right answer?
4Fe (s) + 3 O2 (g) → 2 Fe2O3 (s)
A. What is the oxidation number for each item in this equation?
Questions




Biology, 09.07.2019 20:00

Social Studies, 09.07.2019 20:00

Mathematics, 09.07.2019 20:00





History, 09.07.2019 20:00

Mathematics, 09.07.2019 20:00



Physics, 09.07.2019 20:00


Physics, 09.07.2019 20:00


History, 09.07.2019 20:00

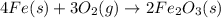
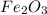 is +3.
is +3. 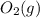 is 0 as it is present in its elemental state.
is 0 as it is present in its elemental state.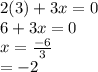
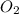 as a decrease in its oxidation state is occurring. So,
as a decrease in its oxidation state is occurring. So, 


