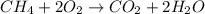Chemistry, 07.06.2021 09:00 malikfitzgerald1910
The complete combustion of methane can be seen in the equation: CH4 + 2 points
202 - CO2 + 2H2O If 50 g of methane are reacted completely, which
statement describes the reaction? *
O The total mass of the reactants is 50 g.
O The total mass of products is 50 g.
O The total mass of products is less than 50 g.
The total mass of reactants is greater than 50 g.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

You know the right answer?
The complete combustion of methane can be seen in the equation: CH4 + 2 points
202 - CO2 + 2H2O If...
Questions


Computers and Technology, 02.03.2021 02:50

Mathematics, 02.03.2021 02:50

Mathematics, 02.03.2021 02:50



Mathematics, 02.03.2021 02:50


Social Studies, 02.03.2021 02:50

Mathematics, 02.03.2021 02:50

Mathematics, 02.03.2021 02:50



Social Studies, 02.03.2021 02:50

Mathematics, 02.03.2021 02:50

Mathematics, 02.03.2021 02:50




Mathematics, 02.03.2021 02:50