
Chemistry, 05.06.2021 22:50 Kaysofine11icloudcom
What is the limiting reactant in a reaction where 10.0 mol of iron is treated with 12.0 mol of bromine? The product that forms is FeBr3. First, write and balance the chemical equation. Next, calculate the moles of FeBr3 that can be made from 10.0 mol of Fe. Then calculate the moles of FeBr3 that can be made from 12.0 mol of bromine. The smaller amount of FeBr3 reveals the limiting reactant.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Naoki's bicycle has a mass of 10 kg. if naoki sits on her bicycle and starts pedaling with a force of 168 n, causing an acceleration of 2.8 m/s2, what is naoki's mass?
Answers: 1

Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1


Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
What is the limiting reactant in a reaction where 10.0 mol of iron is treated with 12.0 mol of bromi...
Questions

History, 05.11.2020 19:10

History, 05.11.2020 19:10


Mathematics, 05.11.2020 19:10


Mathematics, 05.11.2020 19:10

Mathematics, 05.11.2020 19:10

Social Studies, 05.11.2020 19:10







English, 05.11.2020 19:10


English, 05.11.2020 19:10



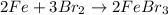
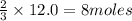 of iron
of iron

