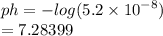Chemistry, 03.06.2021 20:50 michellen2020
What is the PH of a solution with a concentration of 5.2 x 10-8 M H3O?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

You know the right answer?
What is the PH of a solution with a concentration of 5.2 x 10-8 M H3O?...
Questions

Geography, 23.09.2019 21:00



History, 23.09.2019 21:00





Mathematics, 23.09.2019 21:00


History, 23.09.2019 21:00

History, 23.09.2019 21:00



Mathematics, 23.09.2019 21:00

Social Studies, 23.09.2019 21:00

History, 23.09.2019 21:00

Mathematics, 23.09.2019 21:00


Mathematics, 23.09.2019 21:00

![pH = - log [ { H_3O}^{+}]](/tpl/images/1361/6277/1a2ed.png)