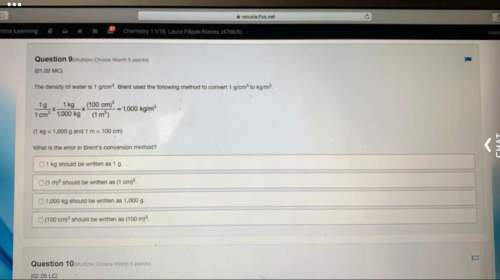
At standard temperature and pressure (STP), 1 mole of any gas occupies:
a)22.4 L of volume
b)...

Chemistry, 01.06.2021 16:40 rhussein6452
At standard temperature and pressure (STP), 1 mole of any gas occupies:
a)22.4 L of volume
b)1 gram of mass
c)22.4 grams of mass
d)1 L of volume

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following methods uses the decay of atomic particles in an object to find its exact age? a. fossil dating b. geologic dating c. radioactive dating d. relative dating
Answers: 1

Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1

Chemistry, 23.06.2019 06:00
Nthis lab, you will do experiments to identify types of changes. using the question format you learned (shown above), write an investigative question that you can answer by doing these experiments
Answers: 3
You know the right answer?
Questions


Mathematics, 15.10.2019 03:00

English, 15.10.2019 03:00

Computers and Technology, 15.10.2019 03:00



Mathematics, 15.10.2019 03:00




English, 15.10.2019 03:00

Mathematics, 15.10.2019 03:00

History, 15.10.2019 03:00

Physics, 15.10.2019 03:00

Mathematics, 15.10.2019 03:00



Health, 15.10.2019 03:00