
An investigation is conducted into how the mass of magnesium metal reacting with hydrochloric acid affects the amount of hydrogen gas produced.
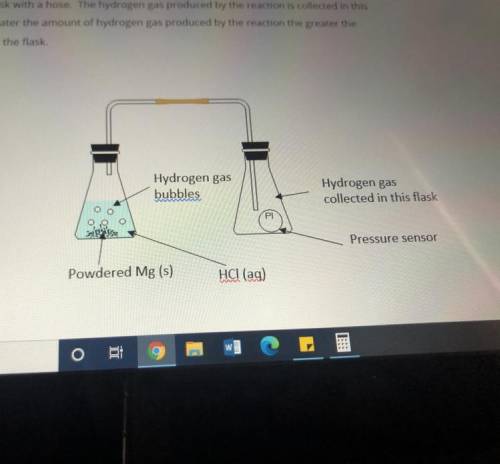
Masses of 0.10g, 0.20g, 0.30g and 0.40g of powdered Mg metal are reacted with hydrochloric acid(HCl). The conical flask containing the reaction mixture of Mg and HCl is connected to another conical flask with a hose. The hydrogen gas produced by the reaction is collected in this conical flask. The greater the amount of hydrogen gas produced by the reaction the greater the pressure of the gas in the flask.
A) what is the independent variable:
B) what is the dependent variable:
C) write a hypothesis for this investigation:
D) give 2 variables that should have been controlled for this investigation:
thank you sm!!!

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3


Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
You know the right answer?
An investigation is conducted into how the mass of magnesium metal reacting with hydrochloric acid a...
Questions


Mathematics, 03.02.2020 18:01

Mathematics, 03.02.2020 18:01

Mathematics, 03.02.2020 18:01


Chemistry, 03.02.2020 18:01

Biology, 03.02.2020 18:01

Spanish, 03.02.2020 18:01

Biology, 03.02.2020 18:01

English, 03.02.2020 18:01

Physics, 03.02.2020 18:01


Mathematics, 03.02.2020 18:01


Mathematics, 03.02.2020 18:01


History, 03.02.2020 18:01

Social Studies, 03.02.2020 18:01




