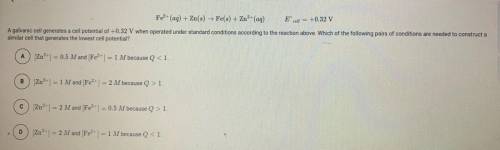Chemistry, 30.05.2021 04:30 cuppykittyy
A galvanic cell generates a cell potential of 0.32V when operated under standard conditions according to the reaction above. Which conditions are needed to construct a similar cell that generates the lowest cell potential?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 07:00
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
You know the right answer?
A galvanic cell generates a cell potential of 0.32V when operated under standard conditions accordin...
Questions


Biology, 02.02.2021 16:10


Mathematics, 02.02.2021 16:10

Mathematics, 02.02.2021 16:20

Physics, 02.02.2021 16:20

Mathematics, 02.02.2021 16:20

Mathematics, 02.02.2021 16:20


Mathematics, 02.02.2021 16:20



Social Studies, 02.02.2021 16:20



Mathematics, 02.02.2021 16:20

Physics, 02.02.2021 16:20