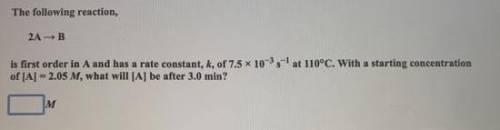Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1
You know the right answer?
Please help with this quickly.
...
...
Questions


Mathematics, 06.10.2019 06:20

Mathematics, 06.10.2019 06:20

Social Studies, 06.10.2019 06:20

Mathematics, 06.10.2019 06:20


Mathematics, 06.10.2019 06:20

Mathematics, 06.10.2019 06:20

Spanish, 06.10.2019 06:20



Chemistry, 06.10.2019 06:20

Business, 06.10.2019 06:20

Arts, 06.10.2019 06:20

History, 06.10.2019 06:20

Biology, 06.10.2019 06:20



Mathematics, 06.10.2019 06:20

English, 06.10.2019 06:20