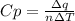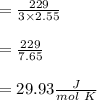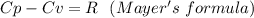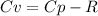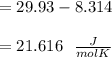Chemistry, 27.05.2021 05:10 genyjoannerubiera
When 229 J of energy is supplied as heat at constant pressure to 3.0mol Ar(g) the temperature of the sample increases by 2.55K. Calculate the molar heat capacities at constant volume and constant pressure of the gas.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1

Chemistry, 23.06.2019 14:00
Which is not true regarding reaction rates? (2 points) catalysts are not used up in the reaction. catalysts speed up reactions by lowering the activation energy. reaction rates decrease as the concentration of reactants decrease. during reactions, concentrations of all reactants decrease at the same rate.
Answers: 1

You know the right answer?
When 229 J of energy is supplied as heat at constant pressure to 3.0mol Ar(g) the temperature of the...
Questions

Mathematics, 21.01.2021 22:10


Business, 21.01.2021 22:10



Mathematics, 21.01.2021 22:10

Mathematics, 21.01.2021 22:10



Computers and Technology, 21.01.2021 22:10

Mathematics, 21.01.2021 22:10




Mathematics, 21.01.2021 22:10





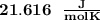 ".
".