Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1
You know the right answer?
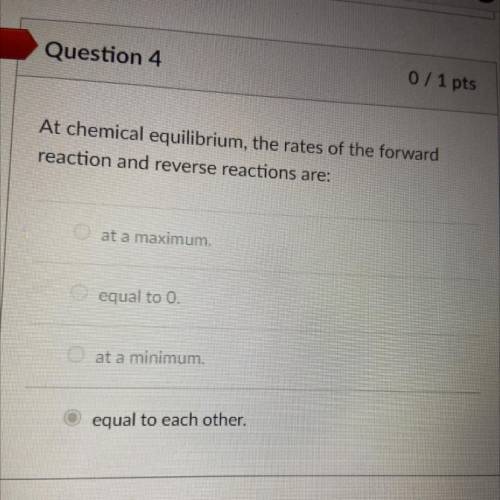
Help me pass this test my last day of school is may 26 if you don’t mind I have more questions :)
Questions

Mathematics, 29.10.2020 06:10

Mathematics, 29.10.2020 06:10

Spanish, 29.10.2020 06:10

English, 29.10.2020 06:10

Physics, 29.10.2020 06:10




Mathematics, 29.10.2020 06:20


Mathematics, 29.10.2020 06:20

Mathematics, 29.10.2020 06:20


Biology, 29.10.2020 06:20


Mathematics, 29.10.2020 06:20

Mathematics, 29.10.2020 06:20


History, 29.10.2020 06:20