
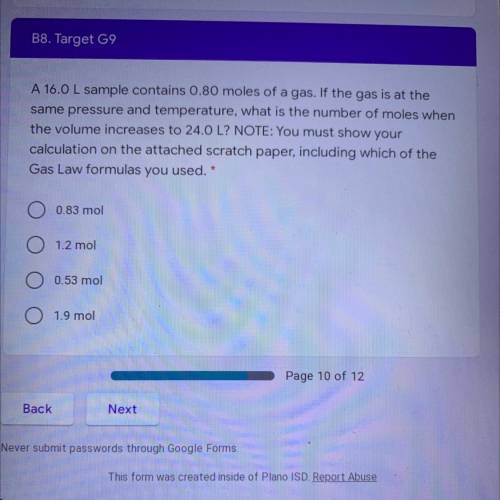
A 16.0 L sample contains 0.80 moles of a gas. If the gas is at the
same pressure and temperature, what is the number of moles when
the volume increases to 24.0 L? NOTE: You must show your
calculation on the attached scratch paper, including which of the
Gas Law formulas you used. *
A. 0.83 mol
B. 1.2 mol
C. 0.53 mol
D. 1.9 mol
(Please show your work)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 06:00
Why is permeable soil best for plants that need a lot of drainage?
Answers: 1

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1
You know the right answer?
A 16.0 L sample contains 0.80 moles of a gas. If the gas is at the
same pressure and temperature, w...
Questions




Health, 24.06.2019 11:40

History, 24.06.2019 11:40




English, 24.06.2019 11:40

English, 24.06.2019 11:40




Chemistry, 24.06.2019 11:40


Geography, 24.06.2019 11:40


Mathematics, 24.06.2019 11:40


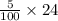 = 120/100= 12/10= 1.2 Mole ( Ans )
= 120/100= 12/10= 1.2 Mole ( Ans )

