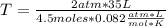Chemistry, 21.05.2021 08:20 Jennifer16253
at what temperature will the mass of 198g of carbon dioxide gas at 2 atmospheric pressure take up to a volume of 35l? (gas constant= 0.0821L atm.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1

Chemistry, 23.06.2019 12:30
An atom holds 7 electrons. use orbital notation to model the probable location of its electrons. an atom hold 22 electrons. use orbital notation to model the probable location of its electrons. an atom holds 17 electrons. use orbital notation to model the probable location of its electrons.
Answers: 1

Chemistry, 23.06.2019 13:30
How many ammonium ions and how many sulfate ions are present in a 0.270 mol sample of ?
Answers: 1
You know the right answer?
at what temperature will the mass of 198g of carbon dioxide gas at 2 atmospheric pressure take up to...
Questions

Business, 19.02.2020 22:53




Biology, 19.02.2020 22:53

English, 19.02.2020 22:53









Mathematics, 19.02.2020 22:54

Computers and Technology, 19.02.2020 22:54



Mathematics, 19.02.2020 22:54

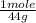 = 4.5 moles (where 44 g/mole is the molar mass of carbon dioxide)R= 0.082
= 4.5 moles (where 44 g/mole is the molar mass of carbon dioxide)R= 0.082 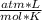 T= ?
T= ?