Chemistry, 20.05.2021 19:40 nicholasferrell
The initial temperature of the water in a constant-pressure calorimeter is
14°C. A reaction takes place in the calorimeter, and the temperature rises
to 87°C. The calorimeter contains 254 g of water, which has a specific heat
of 4.18 J/(g.°C). Calculate the enthalpy change during this reaction. *

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1


Chemistry, 23.06.2019 02:00
The bohr model of the atom explained why emission spectra are discrete. it could also be used to explain the photoelectric effect. which is a correct explanation of the photoelectric effect according to the model?
Answers: 3
You know the right answer?
The initial temperature of the water in a constant-pressure calorimeter is
14°C. A reaction takes p...
Questions


English, 20.09.2019 06:50



English, 20.09.2019 06:50


Mathematics, 20.09.2019 06:50

English, 20.09.2019 06:50

Mathematics, 20.09.2019 06:50


English, 20.09.2019 06:50


Mathematics, 20.09.2019 06:50



Biology, 20.09.2019 06:50

History, 20.09.2019 06:50

English, 20.09.2019 06:50


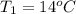 ,
, 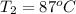
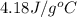
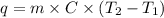
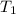 = initial temperature
= initial temperature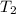 = final temperature
= final temperature