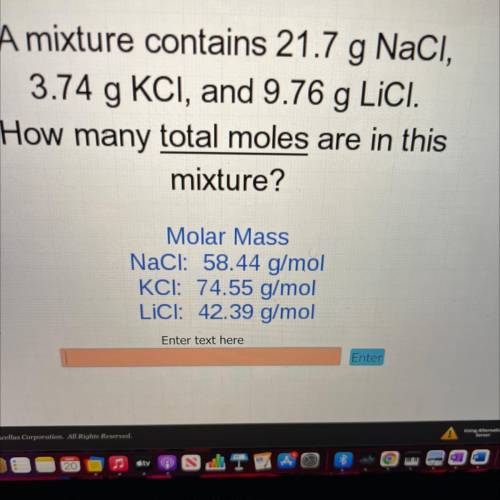
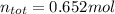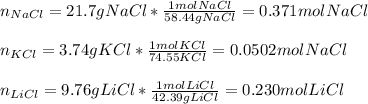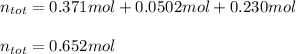A mixture contains 21.7 g NaCl,
3.74 g KCI, and 9.76 g Lici.
How many total moles are in this...


Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 00:50
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1


Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2
You know the right answer?
Questions





Mathematics, 13.12.2019 20:31



Biology, 13.12.2019 20:31


History, 13.12.2019 20:31




Mathematics, 13.12.2019 20:31

Mathematics, 13.12.2019 20:31



Business, 13.12.2019 20:31

Business, 13.12.2019 20:31