
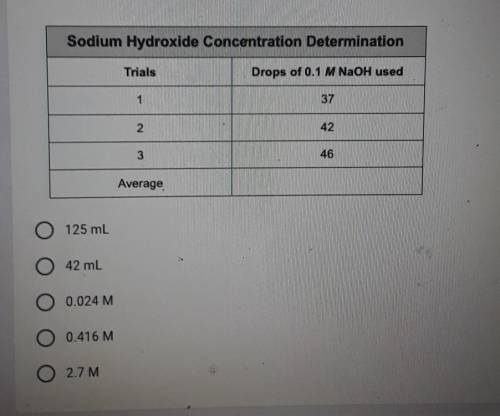
り A drop wise titration of an unknown concentration of vinegar with 0.1 M sodium hydroxide. 10 drops vinegar were added to the well plate along with a drop of the indicator bromothymol blue. The drops of NaOH required were recorded in the data table below. What was the concentration of vinegar based on this data?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2


Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1
You know the right answer?
り A drop wise titration of an unknown concentration of vinegar with 0.1 M sodium hydroxide. 10 drops...
Questions

Social Studies, 11.01.2020 01:31


Social Studies, 11.01.2020 01:31



Social Studies, 11.01.2020 01:31

Social Studies, 11.01.2020 01:31




Social Studies, 11.01.2020 01:31


Physics, 11.01.2020 01:31

Social Studies, 11.01.2020 01:31








