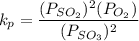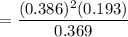Chemistry, 19.05.2021 19:10 sarahaziz9526
A student ran the following reaction in the laboratory at 1080 K: 2SO3(g) 2SO2(g) + O2(g) When she introduced SO3(g) at a pressure of 0.948 atm into a 1.00 L evacuated container, she found the equilibrium partial pressure of SO3(g) to be 0.369 atm. Calculate the equilibrium constant, Kp, she obtained for this reaction. Kp =

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 10:00
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1
You know the right answer?
A student ran the following reaction in the laboratory at 1080 K: 2SO3(g) 2SO2(g) + O2(g) When she i...
Questions

Geography, 14.10.2019 02:00


Mathematics, 14.10.2019 02:00


History, 14.10.2019 02:00

History, 14.10.2019 02:00



Mathematics, 14.10.2019 02:00

Physics, 14.10.2019 02:00

History, 14.10.2019 02:00





Mathematics, 14.10.2019 02:00


Physics, 14.10.2019 02:00


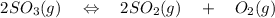
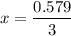
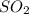 = 0.193 x 2
= 0.193 x 2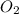 = 0.193 atm
= 0.193 atm