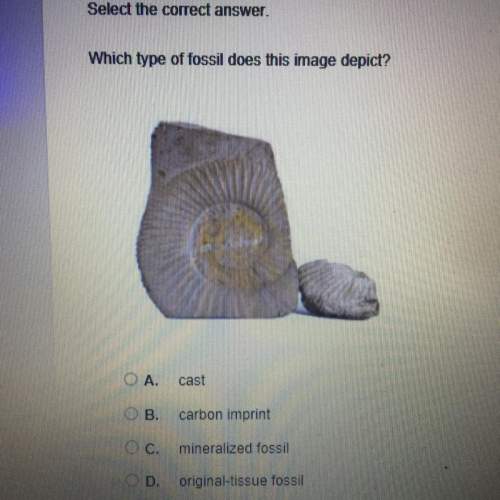
178

Chemistry, 19.05.2021 19:00 gatorboy1788
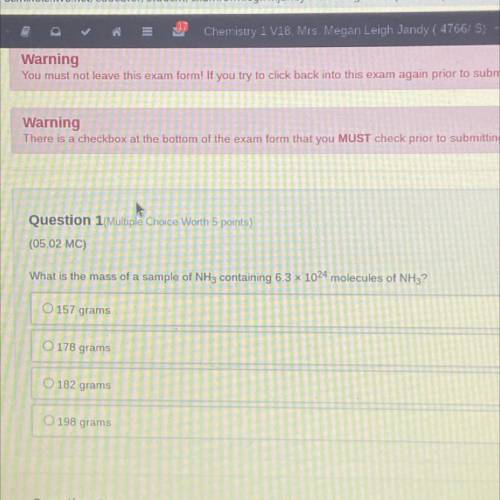
What is the mass of a sample f NH3 containing 6.3 x 10^23 molecules of NH3?
157
178
182
198

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1
You know the right answer?
What is the mass of a sample f NH3 containing 6.3 x 10^23 molecules of NH3?
157
178
178
Questions

History, 19.05.2020 03:08

Mathematics, 19.05.2020 03:08


History, 19.05.2020 03:08

Social Studies, 19.05.2020 03:08

Geography, 19.05.2020 03:08

Mathematics, 19.05.2020 03:08


Computers and Technology, 19.05.2020 03:08



English, 19.05.2020 03:08

Advanced Placement (AP), 19.05.2020 03:08

History, 19.05.2020 03:08


Mathematics, 19.05.2020 03:08

Mathematics, 19.05.2020 03:08


Geography, 19.05.2020 03:08