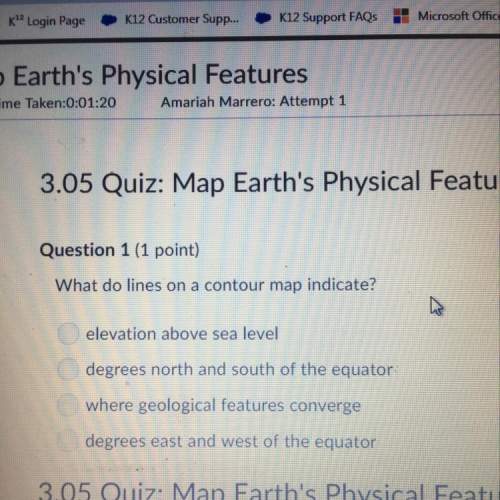
A 0.27-Liter sample of gas has a
pressure of 1.00 atm. What pressure
would have to be applied...

Chemistry, 17.05.2021 20:20 datgamer13
A 0.27-Liter sample of gas has a
pressure of 1.00 atm. What pressure
would have to be applied to the gas to
compress its volume to 0.10 Liter?
Boyle's Law

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1


Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1
You know the right answer?
Questions



History, 24.02.2021 04:00



Mathematics, 24.02.2021 04:00




Computers and Technology, 24.02.2021 04:00

History, 24.02.2021 04:00


Mathematics, 24.02.2021 04:00

Chemistry, 24.02.2021 04:00

English, 24.02.2021 04:00


Mathematics, 24.02.2021 04:00

Business, 24.02.2021 04:00

Mathematics, 24.02.2021 04:00

Social Studies, 24.02.2021 04:00