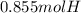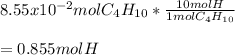Chemistry, 16.05.2021 04:20 tamekiablair502
Determine the number of moles of hydrogen atoms in each of the following. 8.55×10^-2 mol C4H10

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 14:30
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1


Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
Determine the number of moles of hydrogen atoms in each of the following.
8.55×10^-2 mol C4H10...
Questions

English, 03.11.2020 19:20


Mathematics, 03.11.2020 19:20


Mathematics, 03.11.2020 19:20



Computers and Technology, 03.11.2020 19:20

Mathematics, 03.11.2020 19:20

Chemistry, 03.11.2020 19:20



Biology, 03.11.2020 19:20

Mathematics, 03.11.2020 19:20


Biology, 03.11.2020 19:20

History, 03.11.2020 19:20


Mathematics, 03.11.2020 19:20

Chemistry, 03.11.2020 19:20