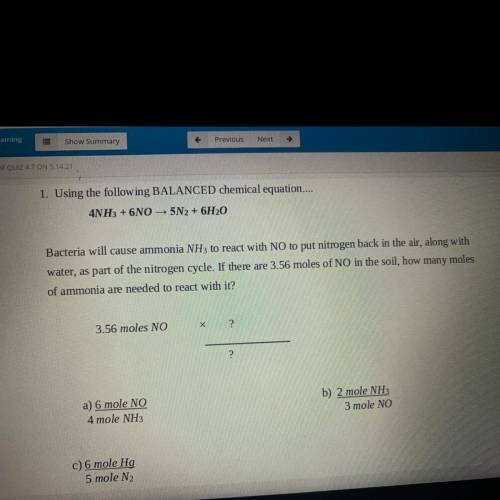
1. Using the following BALANCED chemical equation.
4NH3 + 6NO - 5N2 + 6H20
Bacteria will caus...

Chemistry, 14.05.2021 17:40 hmontalvo22
1. Using the following BALANCED chemical equation.
4NH3 + 6NO - 5N2 + 6H20
Bacteria will cause ammonia NH3 to react with NO to put nitrogen back in the air, along with
water, as part of the nitrogen cycle. If there are 3.56 moles of NO in the soil, how many moles
of ammonia are needed to react with it?
3.56 moles NO
2
2
a) 6 mole NO
4 mole NH3
b) 2 mole NH3
3 mole NO
c) 6 mole Hg
5 mole N2

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
You know the right answer?
Questions

History, 29.10.2020 17:40


Mathematics, 29.10.2020 17:40

History, 29.10.2020 17:40

Mathematics, 29.10.2020 17:40

Chemistry, 29.10.2020 17:40

Mathematics, 29.10.2020 17:40



English, 29.10.2020 17:40

Mathematics, 29.10.2020 17:40


Mathematics, 29.10.2020 17:40

Mathematics, 29.10.2020 17:40

English, 29.10.2020 17:40

Physics, 29.10.2020 17:40

English, 29.10.2020 17:40

Biology, 29.10.2020 17:40


Mathematics, 29.10.2020 17:40



