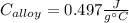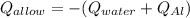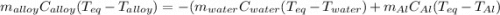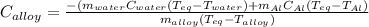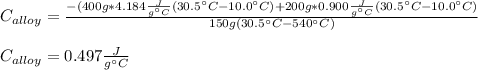Chemistry, 13.05.2021 20:40 ptanner706
A 0.150-kg sample of a metal alloy is heated at 540 Celsius an then plunged into a 0.400-kg of water at 10.0 Celsius, which is contained in a 0.200-kg aluminum calorimeter cup. The final temperature of the system is 30.5 Celsius. What is the specific heat of the metal alloy in J/Kg. Celsius

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2

Chemistry, 23.06.2019 01:00
Aman applies a force of 500n to push a truck 100m down the street how much does he do?
Answers: 1

Chemistry, 23.06.2019 05:30
What is the body’s main processing system? it uses input from various parts to control voluntary and involutiontary movement. it’s composed of two main parts-the brain and spinal cord. a. nbs b.cns c. ans d. pns
Answers: 1
You know the right answer?
A 0.150-kg sample of a metal alloy is heated at 540 Celsius an then plunged into a 0.400-kg of water...
Questions


Social Studies, 27.04.2021 20:30


Mathematics, 27.04.2021 20:30




Mathematics, 27.04.2021 20:30


Mathematics, 27.04.2021 20:30

Mathematics, 27.04.2021 20:30



Chemistry, 27.04.2021 20:30

Mathematics, 27.04.2021 20:30

History, 27.04.2021 20:30


Advanced Placement (AP), 27.04.2021 20:30


Computers and Technology, 27.04.2021 20:30