
Chemistry, 13.05.2021 19:10 bethyboop1820
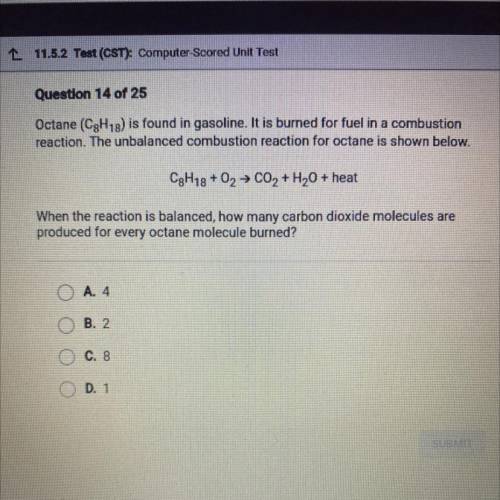
Octane (C8H18) is found in gasoline. It is burned for fuel in a combustion
reaction. The unbalanced combustion reaction for octane is shown below.
C8h18+ O2 → C02 + H2O + heat
When the reaction is balanced, how many carbon dioxide molecules are
produced for every octane molecule burned?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 22.06.2019 14:30
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2


Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2
You know the right answer?
Octane (C8H18) is found in gasoline. It is burned for fuel in a combustion
reaction. The unbalanced...
Questions

Mathematics, 10.02.2020 16:45


Mathematics, 10.02.2020 16:45

Mathematics, 10.02.2020 16:45

Arts, 10.02.2020 16:46


Mathematics, 10.02.2020 16:49



Social Studies, 10.02.2020 16:52










English, 10.02.2020 16:53



