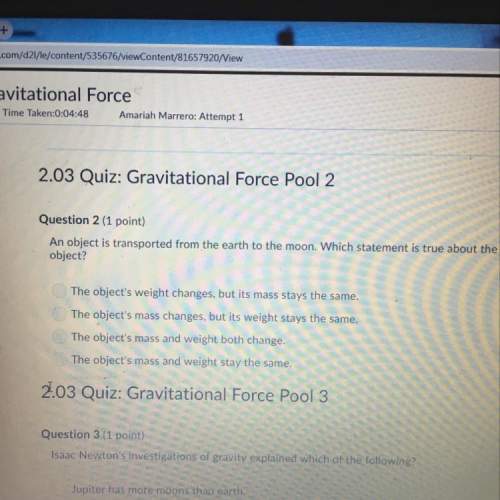
Chemistry, 12.05.2021 17:40 mariahbugg7
Write thermochemical equations for the formation of the following solutions, given their molar
heats of solution.
1. solid sodium cyanide, AH. din = 1.21 kJ/mol
2. solid rubidium hydroxide, AH dn = -62.34 kJ/mol
Rb2O(S)+H20-2RbOH(aq)+
I
3. solid potassium bromide, AH..in = 19.87 kJ/mol
Exceeds
Use the thermochemical equations you constructed in the "Meets" section to calculate the


Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2
You know the right answer?
Write thermochemical equations for the formation of the following solutions, given their molar
heat...
Questions

Mathematics, 06.10.2019 18:30


History, 06.10.2019 18:30

History, 06.10.2019 18:30



Biology, 06.10.2019 18:30

History, 06.10.2019 18:30



Social Studies, 06.10.2019 18:30

History, 06.10.2019 18:30

History, 06.10.2019 18:30


Mathematics, 06.10.2019 18:30


Mathematics, 06.10.2019 18:30



History, 06.10.2019 18:30