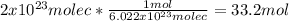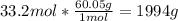Chemistry, 12.05.2021 01:20 jaylenmiller437
En una botella hay 2x10 25 moléculas de vinagre puro ¿Cuántos mol y cuántos gramos de esta sustancia habrá en la botella?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

You know the right answer?
En una botella hay 2x10 25 moléculas de vinagre puro ¿Cuántos mol y cuántos gramos de esta sustancia...
Questions







History, 05.05.2020 00:45

Mathematics, 05.05.2020 00:45

Mathematics, 05.05.2020 00:45


History, 05.05.2020 00:45


Mathematics, 05.05.2020 00:45

World Languages, 05.05.2020 00:45

English, 05.05.2020 00:45