Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 22.06.2019 14:30
Ahypothesis must be testable and falsifiable to be considered scientific a. trueb. false
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
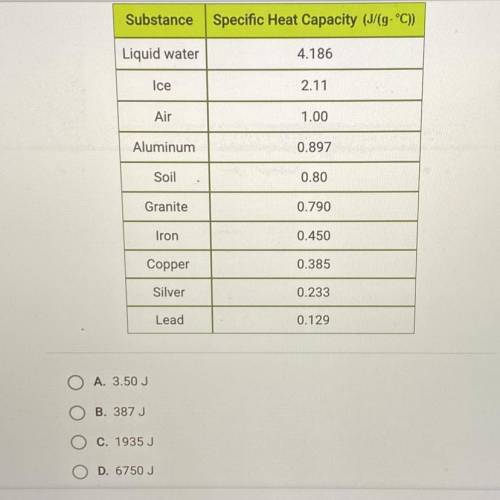
How much energy is required to raise the temperature of 3 kg of lead from 15°C to 20°C? Use the tabl...
Questions

English, 30.04.2021 23:30


English, 30.04.2021 23:30


Computers and Technology, 30.04.2021 23:30


Mathematics, 30.04.2021 23:30


Mathematics, 30.04.2021 23:30



Mathematics, 30.04.2021 23:30


Mathematics, 30.04.2021 23:30

Mathematics, 30.04.2021 23:30

Computers and Technology, 30.04.2021 23:30



Mathematics, 30.04.2021 23:30