
Chemistry, 08.05.2021 08:10 sparky1234
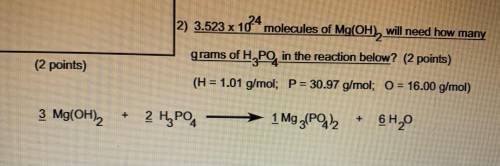
3.523 x 10^24 molecules of Mg(OH2) will need how many grams of H3PO4 in the reaction below
H=1.01 g/mol
P=30.97 g/mol
O=16.00 g/mol
3 Mg(OH)2 + 2 H3PO4 -> 1 Mg3 (PO4 )2 + 6 H2O

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2
You know the right answer?
3.523 x 10^24 molecules of Mg(OH2) will need how many grams of H3PO4 in the reaction below
H=1.01...
Questions

Physics, 25.01.2022 02:00

History, 25.01.2022 02:00


Arts, 25.01.2022 02:00

Mathematics, 25.01.2022 02:00

English, 25.01.2022 02:00






Business, 25.01.2022 02:00

History, 25.01.2022 02:00



Mathematics, 25.01.2022 02:00

Computers and Technology, 25.01.2022 02:00

Mathematics, 25.01.2022 02:00


Mathematics, 25.01.2022 02:00



