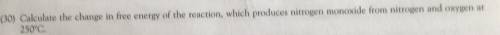Please help me! You’re getting 16 for answering this!
...

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:50
What type of reaction is illustrated? 2c12o5 = 2cl2 + 502
Answers: 2

Chemistry, 21.06.2019 15:40
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.
Answers: 3

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

You know the right answer?
Questions


Computers and Technology, 29.08.2019 01:20


Computers and Technology, 29.08.2019 01:20

Computers and Technology, 29.08.2019 01:20

History, 29.08.2019 01:20




Mathematics, 29.08.2019 01:20

English, 29.08.2019 01:20

Mathematics, 29.08.2019 01:20

Mathematics, 29.08.2019 01:20

Mathematics, 29.08.2019 01:20