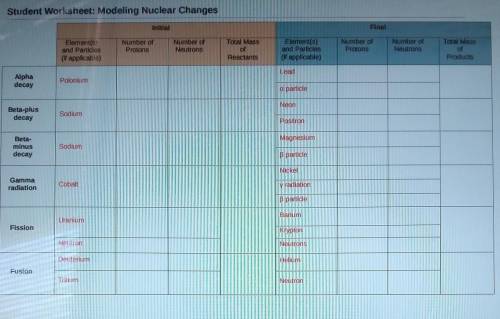
*PLEASE HELP**NEED HELP SOON*
Please fill out the table
...

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2


Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
Questions

Computers and Technology, 01.01.2021 22:50



French, 01.01.2021 22:50

Computers and Technology, 01.01.2021 22:50


SAT, 01.01.2021 22:50

Medicine, 01.01.2021 22:50

Physics, 01.01.2021 22:50

Geography, 01.01.2021 22:50


Mathematics, 01.01.2021 22:50

Mathematics, 01.01.2021 22:50

Mathematics, 01.01.2021 22:50

English, 01.01.2021 22:50

Physics, 01.01.2021 22:50

Biology, 01.01.2021 22:50

Biology, 01.01.2021 22:50

Mathematics, 01.01.2021 22:50