Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2
You know the right answer?
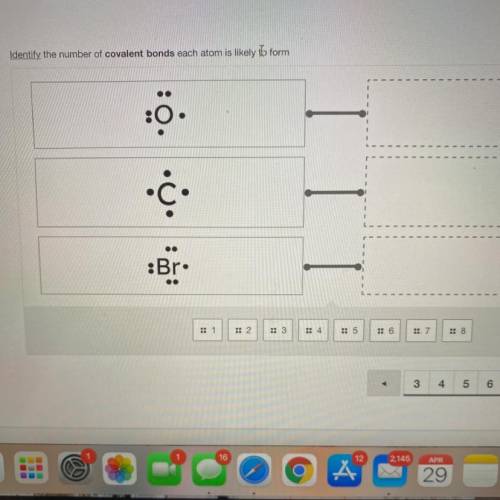
Help please
i really need the answer please
...
i really need the answer please
...
Questions



Mathematics, 22.11.2019 20:31

History, 22.11.2019 20:31



History, 22.11.2019 20:31

Biology, 22.11.2019 20:31

Mathematics, 22.11.2019 20:31

Mathematics, 22.11.2019 20:31

Mathematics, 22.11.2019 20:31


Mathematics, 22.11.2019 20:31

Mathematics, 22.11.2019 20:31


Mathematics, 22.11.2019 20:31

History, 22.11.2019 20:31

Mathematics, 22.11.2019 20:31

Mathematics, 22.11.2019 20:31