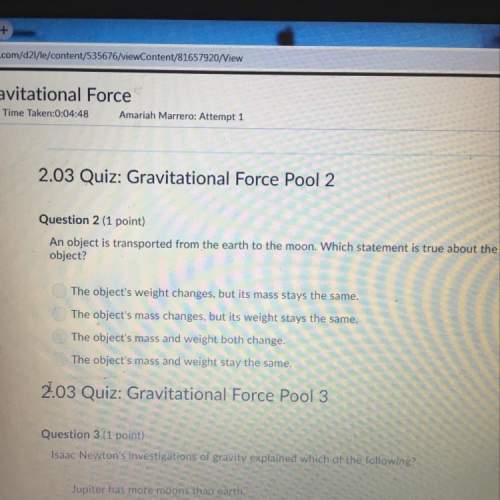
Reaction intermediates differ from activated complexes in that A. they are stable molecules with normal bonds and are frequently isolated. B. they are molecules with normal bonds rather than partial bonds and can occasionally be isolated. C. they are intermediate structures which have characteristics of both reactants and products. D. they are unstable and can never be isolated. E. all reactions involve reaction intermediates, but not all have activated complexes.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:30
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2
You know the right answer?
Reaction intermediates differ from activated complexes in that A. they are stable molecules with nor...
Questions


World Languages, 22.02.2021 22:40



Mathematics, 22.02.2021 22:40


Mathematics, 22.02.2021 22:40

Mathematics, 22.02.2021 22:40

Social Studies, 22.02.2021 22:40

Mathematics, 22.02.2021 22:40

Mathematics, 22.02.2021 22:40


Chemistry, 22.02.2021 22:40

History, 22.02.2021 22:40





Biology, 22.02.2021 22:40