Chemistry, 22.04.2021 03:20 lanakay2006
The following energy diagram depicts the energy changes involved for A+B AB
(a) Is the following diagram an exothermic or endothermic reaction?
(b) Explain how you arrived at this conclusion.

Answers: 2


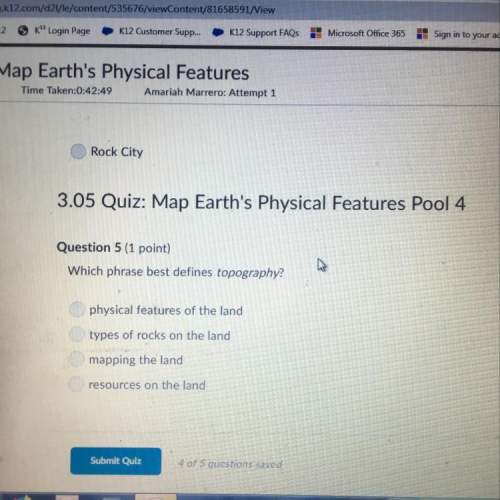
Another question on Chemistry

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2

Chemistry, 23.06.2019 04:31
Which molecules are more strongly attracted to one another -c3h8o molecules that make up liquid rubbing alcohol or ch4 molecules that make up methane gas
Answers: 3

Chemistry, 23.06.2019 04:40
[01.07]what is the answer to the problem: 101 g + 25.01 g + 5.05 g? 131.06 g 131.1 g 131 g 130 g
Answers: 1
You know the right answer?
The following energy diagram depicts the energy changes involved for A+B AB
(a) Is the following d...
Questions

Business, 17.11.2020 19:00

Arts, 17.11.2020 19:00



Computers and Technology, 17.11.2020 19:00

Mathematics, 17.11.2020 19:00


English, 17.11.2020 19:00

English, 17.11.2020 19:00


Mathematics, 17.11.2020 19:00

Biology, 17.11.2020 19:00

Mathematics, 17.11.2020 19:00




Mathematics, 17.11.2020 19:00