
Chemistry, 22.04.2021 01:20 emmanuel180
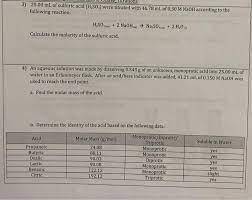
An aqueous solution was made by dissolving 0.543 g of an unknown, monoprotic acid into 25.00 mL of water in an Erlenmeyer flask. After an acid/base indicator was added, 41.21 mL of 0.150 M NaOH was used to reach the end point.
a)Find the molar mass of the acid.
b)Determine the identity of the acid based on the following data:
PLEASE HELP ME

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1


Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
An aqueous solution was made by dissolving 0.543 g of an unknown, monoprotic acid into 25.00 mL of w...
Questions

Social Studies, 18.12.2021 14:00


SAT, 18.12.2021 14:00

Mathematics, 18.12.2021 14:00

Social Studies, 18.12.2021 14:00



Mathematics, 18.12.2021 14:00

Mathematics, 18.12.2021 14:00

Advanced Placement (AP), 18.12.2021 14:00



Social Studies, 18.12.2021 14:00

English, 18.12.2021 14:00



History, 18.12.2021 14:00

Mathematics, 18.12.2021 14:00

Spanish, 18.12.2021 14:00




