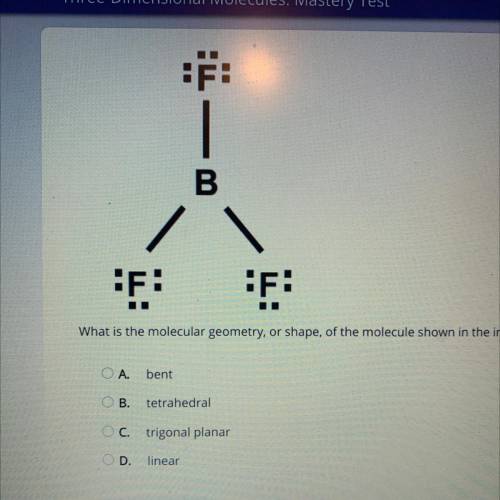
What is the molecular geometry, or shape, of the molecule shown in the image?
A. bent
B. tet...


Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
For the following, determine the type of reaction and then give products.
Answers: 2

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
Questions




Mathematics, 24.02.2021 06:20



Mathematics, 24.02.2021 06:20

History, 24.02.2021 06:20



Mathematics, 24.02.2021 06:20




English, 24.02.2021 06:20


Mathematics, 24.02.2021 06:20

Mathematics, 24.02.2021 06:20

Spanish, 24.02.2021 06:20