Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 07:00
The blackbody curve for a star name zeta is shown below. what is the peak wavelength for this star ?
Answers: 1

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3
You know the right answer?
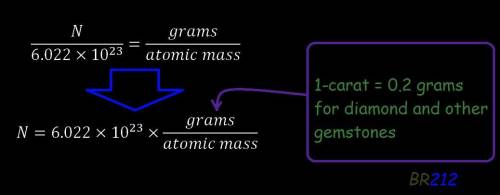
How many carbon atoms are there in a 1.3-carat diamond? diamonds are a form of pure carbon. (1 cara...
Questions


English, 19.11.2020 21:00

Mathematics, 19.11.2020 21:00


Mathematics, 19.11.2020 21:00


Mathematics, 19.11.2020 21:00

Mathematics, 19.11.2020 21:00

Mathematics, 19.11.2020 21:00


Mathematics, 19.11.2020 21:00





Mathematics, 19.11.2020 21:00



Mathematics, 19.11.2020 21:00