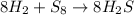Consider the balanced equation below. 8h2 + s8 mc006-1.jpg 8h2s based on the mole ratios, what can most likely be predicted? 1 mol of hydrogen will react with 1 mol of sulfur. 8 mol of hydrogen will react with 1 mol of sulfur. 8 mol of hydrogen will react with 8 mol of sulfur. 16 mol of hydrogen will react with 8 mol of sulfur.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 18:00
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3

Chemistry, 23.06.2019 05:00
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2

Chemistry, 23.06.2019 06:30
Generally, observed behavior that can be formulated into a statement, sometimes mathematical in nature, is called a(n): a. observation. b. measurement. c. theory. d. natural law. e. experiment.
Answers: 2
You know the right answer?
Consider the balanced equation below. 8h2 + s8 mc006-1.jpg 8h2s based on the mole ratios, what can m...
Questions


Mathematics, 08.12.2020 01:00




Mathematics, 08.12.2020 01:00


History, 08.12.2020 01:00


Mathematics, 08.12.2020 01:00

English, 08.12.2020 01:00

Social Studies, 08.12.2020 01:00

Mathematics, 08.12.2020 01:00


Mathematics, 08.12.2020 01:00


English, 08.12.2020 01:00



Advanced Placement (AP), 08.12.2020 01:00