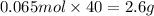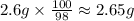Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
You know the right answer?
What mass of naoh is required to prepare 500.00 ml of 0.1300 m naoh stock solution, if the solid nao...
Questions

Mathematics, 16.04.2021 21:20

Mathematics, 16.04.2021 21:20






Social Studies, 16.04.2021 21:20

Mathematics, 16.04.2021 21:20

Mathematics, 16.04.2021 21:20



Arts, 16.04.2021 21:20


History, 16.04.2021 21:20


Social Studies, 16.04.2021 21:20

Mathematics, 16.04.2021 21:20


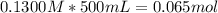
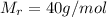 ) needed:
) needed: