
Chemistry, 17.10.2019 03:00 Foxfire5109
How many liters of methane gas (ch4) need to be combusted to produce 8.5 liters of water vapor, if all measurements are taken at the same temperature and pressure? show all of the work used to solve this problem. ch4 (g) + 2o2 (g) yields co2 (g) + 2h2o (g)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2
You know the right answer?
How many liters of methane gas (ch4) need to be combusted to produce 8.5 liters of water vapor, if a...
Questions


Mathematics, 17.07.2021 18:10

English, 17.07.2021 18:10

Mathematics, 17.07.2021 18:10

Mathematics, 17.07.2021 18:20

Mathematics, 17.07.2021 18:20

English, 17.07.2021 18:20


Mathematics, 17.07.2021 18:20

Health, 17.07.2021 18:20

Mathematics, 17.07.2021 18:20

Chemistry, 17.07.2021 18:20

English, 17.07.2021 18:20


Computers and Technology, 17.07.2021 18:20



Mathematics, 17.07.2021 18:20


Mathematics, 17.07.2021 18:20

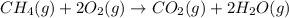
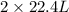 volume of water vapor produced from 22.4 L volume of methane gas
volume of water vapor produced from 22.4 L volume of methane gas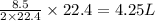 volume of methane gas
volume of methane gas

