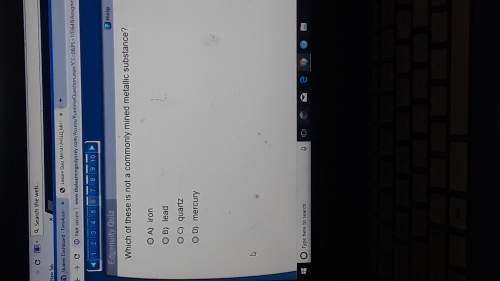
Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1

Chemistry, 22.06.2019 21:20
The organs inside the body and how they function together
Answers: 3


Chemistry, 23.06.2019 06:30
Acertain atom has 22 protons and 19 electrons. this atom loses an electron. the net charge on the atom is now 4+1+01-4-. if this same atom with 22 protons and 19 electrons were to gain 3 electrons, the net charge on the atom would be 3+2+02-3-.
Answers: 1
You know the right answer?
Given the reaction: c6h12o6(s) + 6 o2(g) 6 co2(g) + 6 h2o() how many moles of c6h12o6(s) are needed...
Questions


Mathematics, 20.08.2019 19:00



Physics, 20.08.2019 19:00

Mathematics, 20.08.2019 19:00


Mathematics, 20.08.2019 19:00




Chemistry, 20.08.2019 19:00








Biology, 20.08.2019 19:00