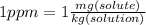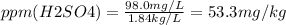Chemistry, 24.11.2019 20:31 brownzackery71
Asolution of sulfuric acid has a concentration of 0.0980 g/l. if the density of the acid is 1.84 g/ml, what is the concentration in ppm?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

You know the right answer?
Asolution of sulfuric acid has a concentration of 0.0980 g/l. if the density of the acid is 1.84 g/m...
Questions






Biology, 12.12.2019 19:31






Mathematics, 12.12.2019 19:31



History, 12.12.2019 19:31

Computers and Technology, 12.12.2019 19:31

Chemistry, 12.12.2019 19:31