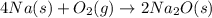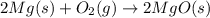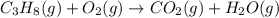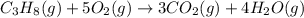Chemistry, 02.12.2019 03:31 itscheesycheedar
Which of the following are balanced equations? check all that apply. a. 4na(s) + o2(g) → 2na2o(s) b. 2mg(s) + o2(g) → 2mgo(s) c. c3h8(g) + o2(g) → co2(g) + h2o(g) d. c3h8(g) + 5o2(g) → 3co2(g) + 4h2o(g

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1
You know the right answer?
Which of the following are balanced equations? check all that apply. a. 4na(s) + o2(g) → 2na2o(s) b...
Questions



Biology, 02.07.2019 02:20

History, 02.07.2019 02:20

Physics, 02.07.2019 02:20


History, 02.07.2019 02:20



Mathematics, 02.07.2019 02:20



Advanced Placement (AP), 02.07.2019 02:20

Biology, 02.07.2019 02:20



Mathematics, 02.07.2019 02:20


Social Studies, 02.07.2019 02:20