Chemistry, 30.08.2019 15:10 princessgabbee8452
Asample of three mixed gases is at 632.0 mmhg. if the partial pressure of co2 is 124.3 mmhg and the partial pressure of n2 is 461.9 mmhg, what is the partial pressure of o2?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Dwayne filled a small balloon with air at 298.5 k. he put the balloon into a bucket of water, and the water level in the bucket increased by 0.54 liter. if dwayne puts the balloon into a bucket of ice water at 273.15 k and waits for the air inside the balloon come to the same temperature, what will the volume of the balloon be? assume the pressure inside the balloon doesn’t change. type the correct answer in the box. express your answer to the correct number of significant figures. the volume of the balloon at 273.15 k is liters.
Answers: 2

Chemistry, 21.06.2019 15:30
Anurse practitioner prepares an injection of promethazine, an antihistamine used to treat allergic rhinitis. if the stock bottle is labeled 25 mg/ml and the order is a dose of 11.0 mg , how many milliliters will the nurse draw up in the syringe?
Answers: 3

Chemistry, 21.06.2019 21:00
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1
You know the right answer?
Asample of three mixed gases is at 632.0 mmhg. if the partial pressure of co2 is 124.3 mmhg and the...
Questions




















Mathematics, 28.11.2019 19:31

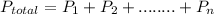
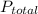 = 632.0 mmhg,
= 632.0 mmhg, 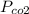 = 124.3 mmhg
= 124.3 mmhg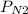 = 461.9
= 461.9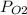 = ?
= ?