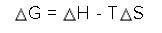Areaction has the following values. calculate δg.
δh = -85.5 kj/mole
δs = 0.133 kj/mole...

Chemistry, 26.08.2019 21:50 jreasley3783
Areaction has the following values. calculate δg.
δh = -85.5 kj/mole
δs = 0.133 kj/mole. k)
temperature = 110°c
-35 kj
-64 kj
-86 kj
-136 kj

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1


Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1
You know the right answer?
Questions

Chemistry, 20.11.2020 17:20

Mathematics, 20.11.2020 17:20


Mathematics, 20.11.2020 17:20

English, 20.11.2020 17:20

Physics, 20.11.2020 17:20

Mathematics, 20.11.2020 17:20

Mathematics, 20.11.2020 17:20






Computers and Technology, 20.11.2020 17:20

Chemistry, 20.11.2020 17:20



Engineering, 20.11.2020 17:20


Social Studies, 20.11.2020 17:20