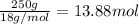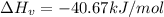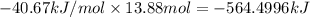Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3

Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 23.06.2019 05:00
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2
You know the right answer?
What is the change in enthalpy when 250 g of water vapor condenses at 100oc? (δhv = 40.67 kj/mol)...
Questions





Chemistry, 18.05.2021 19:40

Physics, 18.05.2021 19:40

Arts, 18.05.2021 19:40



Mathematics, 18.05.2021 19:40

Arts, 18.05.2021 19:40


Mathematics, 18.05.2021 19:40







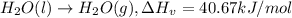
![[tex]H_2O(g)\rightarrow H_2O(l),\Delta H_c=-40.67 kJ/mol](/tpl/images/0262/4002/ebf91.png)