
Chemistry, 22.11.2019 16:31 angelina12386
Combustion analysis of an unknown compound provides the following data: 73.5 grams c, 4.20 grams h, and 72.3 grams cl. what is the percent composition of each element in this compound?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
Combustion analysis of an unknown compound provides the following data: 73.5 grams c, 4.20 grams h,...
Questions

History, 18.06.2020 22:57



Mathematics, 18.06.2020 22:57



Computers and Technology, 18.06.2020 22:57





Mathematics, 18.06.2020 22:57




Mathematics, 18.06.2020 22:57


Chemistry, 18.06.2020 22:57


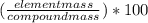
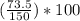 = 49.0%
= 49.0%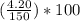 = 2.8%
= 2.8%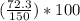 = 48.2%
= 48.2%

