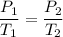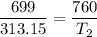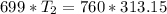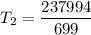Chemistry, 29.09.2019 15:30 kitttimothy55
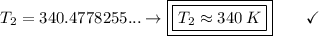
An unknown gas has a pressure of 699.0 mm hg at 40.0 c. what is the temperature at standard pressure?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 03:00
Asample of sea water contains 6.28g of sodium chloride per litre of solution. how many milligrams of sodium chloride would be contained in 15.0ml of this solution?
Answers: 3


Chemistry, 23.06.2019 05:30
Astudent made the lewis dot diagram of a compound as shown. mg is written with two dots shown on its top. an o is written on each side of mg. each o has six dots around it. an arrow is shown from one dot on mg toward the vacant space around the o on the right. another arrow is shown from the other dot on mg toward the vacant space around the o on the left. the title of the art is students lewis dot model. what is the error in the lewis dot diagram? an o atom should transfer all its six electrons to mg because the formula is mgo. both electrons of mg should be transferred to one o atom because the formula is mgo. the electrons should be transferred from each o atom to mg because mg has fewer electrons. the number of dots around mg should be four because it has to transfer two electrons to each o.
Answers: 2
You know the right answer?
An unknown gas has a pressure of 699.0 mm hg at 40.0 c. what is the temperature at standard pressure...
Questions

English, 07.06.2021 17:40

Mathematics, 07.06.2021 17:40

Mathematics, 07.06.2021 17:40






Chemistry, 07.06.2021 17:40


Biology, 07.06.2021 17:40


English, 07.06.2021 17:40

Mathematics, 07.06.2021 17:40

Geography, 07.06.2021 17:40

Mathematics, 07.06.2021 17:40


Chemistry, 07.06.2021 17:40

Mathematics, 07.06.2021 17:40

English, 07.06.2021 17:40